
Urinalysis for diaper-wearing elderly people using a combination of cotton-based diagnostic devices and smartphone-based image analysis
Introduction
Urinary tract infections (UTI) are one of the most common bacterial infections, annually affecting 150 million people worldwide (1). In 2010, the direct and indirect costs of UTI in the United States amounted to approximately $2.3 billion (2). The incidence of UTI in women is far higher than in men. This can be explained by the following two reasons: (I) the shorter female urethra allows colonized bacteria to reach the bladder more easily; and, (II) the female urethral opening is very close to the rectum, which facilitates bacterial migration (3).
Despite the higher incidence among women, the highest prevalence of UTI is actually in the overall elderly population. UTI strongly influences not only community-dwelling older adults but also highly functionally impaired long-term care facilities residents (4). Currently, the diagnosis of UTI is based on the presence of clinical symptoms in combination with positive urine culture results. Unfortunately, the evaluation of urinary symptoms among elderly patients is extremely challenging. First, symptoms of UTI are highly variable in elderly adults, nonspecific to infection. Second, practical problems have occurred in urine sample collection, resulting in inappropriate laboratory results interpretation (5). Furthermore, the diagnosis of symptomatic UTI in older adults with associated disorders such as Alzheimer’s disease, Parkinson’s disease, and stroke is even more challenging (6). Elderly patients suffering from functional or cognitive impairments have great difficulty communicating localized genitourinary symptoms, which can include dysuria, frequency, urgency, suprapubic pain, or hematuria (3).
Given that increased age is associated with higher risk of septic shock, there is an urgent need for a new device for bacteriuria screening and early UTI diagnosis among patients aged 65 years or older (7). We took the approach of designing a device that would facilitate more timely and appropriate antibiotic treatment that could both hasten recovery and counter the trend toward worldwide antibiotic resistance from therapeutic misuse. With innovations in cellphone-based technologies during the last decade, there are considerable advances in point-of-care testing (POCT) devices (8,9). In this study, our aim is to develop an inexpensive, easy to use, rapid, and accurate cotton-based in-vitro diagnostic device (IVD) to detect nitrite concentrations.
Methods
Cellulose-based materials (e.g., paper, cotton and nitrocellulose) have been widely used for both qualitative and quantitative medical diagnostics (e.g., dengue fever and VEGF level) (10,11). Among these naturally-derived materials, cotton provides good flexibility and biocompatibility that facilitate the creation of highly suitable diagnostic devices.
Our diagnostic device comprises three primary parts: (I) a test pad (Whatman chromatography paper); (II) a flow channel made by hydrophobic and hydrophilic cotton (Shiseido cleansing cotton); and, (III) upper and lower plastic packaging substrate (12). Wax printing was used to create complete hydrophobic barriers around test zones. We attached the three part together to make the device. The flow channel allowed fluid to pass removing impurities. This particular urine testing device was integrated directly into a diaper with one side facing the patients’ body, and the reverse side adhered onto the diaper (Figure 1). We converted the colorimetric results to 8-bit grayscale using ImageJ software and defined color intensity as 255– [experiment zone intensity].
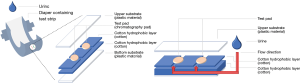
Diapers are useful for sample collection in urinalysis, especially when patients suffer from urinary incontinence. Commonly, the urine sample is collected by catheterization in these cases, contributing to pain and sample contamination (13). By combining a diagnostic device with a diaper, urine samples are collected immediately, reaching test pads through capillary flow. The entire procedure is completed in a single step.
Results
To evaluate the performance of urine test device to detect nitrite, we spotted serially diluted nitrite onto each test zone. The nitrite test color change was from colorless to purple, and the colorimetric reaction was photographed through using a smartphone camera. Figure 2 shows that samples with different nitrite concentrations have a visually distinguishing color intensity that correlates positively to concentration (from 0.156 to 5 µM) in both test paper only and test pad integrated with diaper.

We used a smartphone as a recording device to obtain and analyze the colorimetric-based images when the colorimetric responses occurred in the test pad. The red green blue (RGB) information was then provided accordingly. As a result, we conducted the accurate color analysis by using a comparator card with the known RGB values. We applied our smartphone-based application (APP) as well for timely image analysis (Figure 3). The results became more sensitive and specific once we incorporated the machine learning algorithm.

Discussion
Conventional methods have been widely used to detect bacteria in urine include urine dipsticks and urine culture. The dipsticks are economical with the minimal equipment required; however, they require a relatively large amount of sample (14). The urine culture is considered as a gold standard, to provide the quantitative results but it incurs a fair amount of cost, and time (between 24 and 48 hours). The diagnosis and the following treatment of urinary tract infection are delayed, leading to severe morbidity and mortality.
In comparison to conventional diagnostic methods, our cotton-based device features the following advantages: (I) it detects clinical target in a single step using one device, carrying a lower possibility of clinical sample contamination; (II) it avoids sensitivity and precision issues encountered by the use of unpurified samples in clinical analysis. The design of our device uses chromatography paper and cotton material to create a fluid channel that dilutes concentration and filters unpurified samples before they are absorbed by the chromatography test pad. Notably, this tool may offer significant advantages for diagnostic testing in a variety of arenas including rural/remote areas and long-term health care centers. It can also be used in underdeveloped and developing countries where electricity and laboratory facilities are scarce.
Noting the potential for cross-communication between detection pads designed for testing different biomarkers and resulting interference, we used wax printing to create complete hydrophobic barriers around test zones. We recommend clinical trials to improve sensitivity and specificity as well as possible design optimization that may include the addition of other useful biomarkers.
The color-based signal of our assay can be easily detected by either visual interpretation or smartphone camera (15,16). We have used this approach in a previous study (17). Moving forward, we could optimize the APP for more accurate timely image diagnostics. This device could be optimized through machine learning and potential variance issues with light and focal distance could be solved. The combination of a cotton-based diagnostic device and a smartphone-based APP would significantly contribute to therapeutic advances via early diagnosis, thus improving the quality of life among the elderly across all nations.
In addition to being used among the elderly, this device could be used by infants and pregnant women. Recurrent UTI among infants may be an indicator of congenital urinary tract abnormalities. With the application of our diagnostic device, early diagnosis and initiation of treatment is possible, preventing future renal scarring and end-stage renal disease. Among pregnant women, adaptive changes in the urinary tract during pregnancy predispose them to relatively frequent occurrence of UTIs. Moreover, pregnant women are at higher risk of progressing to upper urinary tract infection (e.g., pyelonephritis) from lower urinary tract infection. UTI during pregnancy may result in severe fetal complications, including preterm labor and low birth weight.
Conclusions
The experimental results indicate that our newly developed diagnostic tool is a useful one-step device for detecting clinical target related to UTI. Because this device is inexpensive, portable, and easy to use, we feel that diapers integrated with this or a similar, optimized diagnostic tool has great potential for development into a commercially viable diagnostic device.
Acknowledgments
Funding: The study was supported by project ‘MOST 108-2745-8-007-001’ of Ministry of Science and Technology, Taiwan.
Footnote
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ht.2019.08.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). In this study, ethics committee approval and informed consent were not considered to be necessary.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Flores-Mireles AL, Walker JN, Caparon M, et al. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol 2015;13:269-84. [Crossref] [PubMed]
- Foxman B. The epidemiology of urinary tract infection. Nat Rev Urol 2010;7:653-60. [Crossref] [PubMed]
- Geerlings SE. Clinical Presentations and Epidemiology of Urinary Tract Infections. Microbiol Spectr 2016;4: [Crossref] [PubMed]
- Nicolle LE. Urinary Tract Infections in the Older Adult. Clin Geriatr Med 2016;32:523-38. [Crossref] [PubMed]
- Detweiler K, Mayers D, Fletcher SG. Bacteruria and Urinary Tract Infections in the Elderly. Urol Clin North Am 2015;42:561-8. [Crossref] [PubMed]
- Rowe TA, Juthani-Mehta M. Diagnosis and management of urinary tract infection in older adults. Infect Dis Clin North Am 2014;28:75-89. [Crossref] [PubMed]
- Hsiao CY, Yang HY, Chang CH, et al. Risk Factors for Development of Septic Shock in Patients with Urinary Tract Infection. Biomed Res Int 2015;2015:717094 [Crossref] [PubMed]
- Vashist SK, Luppa PB, Yeo LY, et al. Emerging Technologies for Next-Generation Point-of-Care Testing. Trends Biotechnol 2015;33:692-705. [Crossref] [PubMed]
- Vashist SK, Mudanyali O. Cellphone-based devices for bioanalytical sciences. Anal Bioanal Chem 2014;406:3263-77. [Crossref] [PubMed]
- Wang HK, Tsai CH, Chen KH, et al. Cellulose-based diagnostic devices for diagnosing serotype-2 dengue fever in human serum. Adv Healthc Mater 2014;3:187-96. [Crossref] [PubMed]
- Hsu MY, Hung YC, Hwang DK, et al. Detection of aqueous VEGF concentrations before and after intravitreal injection of anti-VEGF antibody using low-volume sampling paper-based ELISA. Sci Rep 2016;6:34631. [Crossref] [PubMed]
- Lin SC, Hsu MY, Kuan CM, et al. Cotton-based diagnostic devices. Sci Rep 2014;4:6976. [Crossref] [PubMed]
- Bertao AR, Tao D. Stability of colorimetric results in the detection of urine biomarkers using a paper-based analytical device. Conf Proc IEEE Eng Med Biol Soc 2017;2017:185-8. [PubMed]
- Kayalp D, Dogan K, Ceylan G, et al. Can routine automated urinalysis reduce culture requests? Clin Biochem 2013;46:1285-9. [Crossref] [PubMed]
- Wu MY, Hsu MY, Chen SJ, et al. Point-of-Care Detection Devices for Food Safety Monitoring: Proactive Disease Prevention. Trends Biotechnol 2017;35:288-300. [Crossref] [PubMed]
- Yang K, Peretz-Soroka H, Liu Y, et al. Novel developments in mobile sensing based on the integration of microfluidic devices and smartphones. Lab Chip 2016;16:943-58. [Crossref] [PubMed]
- Tsai TT, Huang CY, Chen CA, et al. Diagnosis of Tuberculosis Using Colorimetric Gold Nanoparticles on a Paper-Based Analytical Device. ACS Sens 2017;2:1345-54. [Crossref] [PubMed]
Cite this article as: Sung WH, Cheng CM. Urinalysis for diaper-wearing elderly people using a combination of cotton-based diagnostic devices and smartphone-based image analysis. Health Technol 2019;3:8.


